RNA-protein interactions
We are interested in the molecular machines that make and break RNA molecules, their evolution and impact on the cellular response against viruses and on genome evolution. Pervasive transcription generates thousands of RNA molecules that do not seem to be immediately useful and are rapidly degraded in the nucleus of eukaryotic cells. Some of these molecules can escape to the cytoplasm, where secondary quality control mechanisms are operating, more or less efficiently to remove these “harmful” RNA from cells. Translation plays a critical role in most cytoplasmic quality control mechanisms, making the study of the molecular interactions involved quite challenging.
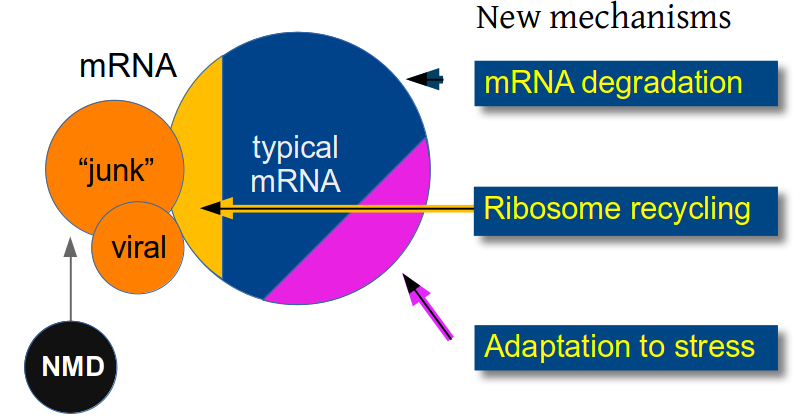
We like to think about the biological questions surrounding mRNA degradation by working on the mechanisms restricting pervasive transcripts (affectionately called "junk" in the figure), those that affect degradation of any other mRNA or those that are specifically involved in "stalled" ribosome situations or shaping the transcriptome response to stress.
The complexity and heterogeneity of mRNA are major barriers in understanding molecular mechanisms involved in their stability and translation. New methods that allow to separate mRNAs with long poly(A) tails from deadenylated ones, new mRNA from older ones and even full-length and capped mRNA from degradation fragments should allow major future discoveries in the field
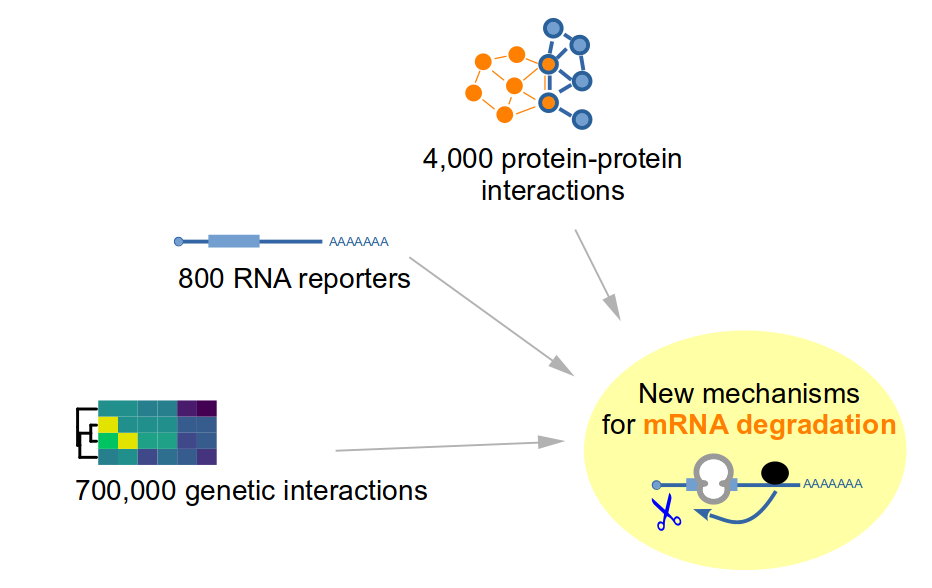
Large-scale experiments can illuminate the factors and mechanisms involved in mRNA degradation. We are using both biochemical techniques and phenotypic screens of thousands of mutants and hundreds of thousands of combinations of mutants to try to understand the relationships between protein complexes and translation in mRNA degradation. Barcoding helped us test hundreds of reporters, leading to new insights into the features affecting mRNA degradation. Previously obtained results are available through interactive web interfaces for genetic interactions and genetic profile similarity (Nucl Acids Res, 2021) and for quantitative proteomics of purified NMD complexes (EmboJ, 2018).
We are particularly interested in understanding the real meaning of correlations that can be found in large-scale data. As an example, mRNA degradation results together with steady-state poly(A) data had strange correlations that could not be explained by any published model for RNA decay. Obtaining our own large scale data together with modeling helped show that deadenylation speed is not a major determinant for mRNA degradation in S. cerevisiae (EmboJ, 2024).